- Co-Promotion of five preventative vaccines for Tdap, hepatitis A, meningococcal disease, measles, and cervical cancer
- With last year’s market size reaching approximately 130 billion won, the two companies will “create synergy through their sales and marketing infrastructures.”
SK bioscience has garnered attention for its world-class technologies and is now strengthening its leading position in the domestic vaccine market.
SK bioscience (CEO Jae-yong Ahn) announced on the 6th that it has signed a co-promotion and distribution agreement for major vaccines with GlaxoSmithKline (GSK), a global pharmaceutical company, to expand its share in the local vaccine market.
Under the joint contract, both SK bioscience and GSK will sell and distribute five GSK vaccines in the Korean market, including vaccines such as “Boostrix” for Tdap (Tetanus, Diphtheria, and Pertussis), “Menveo” for meningococcal disease, “Havrix 1440” for hepatitis A, “Priorix” for measles, mumps, and rubella, and “Cervarix” for cervical cancer.
As the domestic frontrunner in the vaccine market, SK bioscience will be using its solidly established sales network to promote “Boostrix,” “Menveo,” “Havrix 1440,” and “Priorix” in the adult market, while promoting “Cervarix” in all segments including the children’s market.
GSK plans to concentrate its marketing abilities on the vaccine market for infants and children, while expanding its share in the adult vaccine market through co-promotion with SK bioscience.
The market for the five vaccines was estimated at 128 billion won as of last year and is expected to continue growing due to the increased awareness of infectious diseases amid the COVID-19 pandemic.
Boostrix is indicated for immunization of adolescents and adults over the age of 10, and is the only domestic Tdap vaccine that can be given to elderly persons over 65 years of age. Available to the widest range of age groups, Menveo is the only meningococcal vaccine that can be given to infants beginning at 2 months and older.
The world’s first hepatitis A vaccine, Havrix, is sold in over 100 countries, while Cervarix, recognized worldwide for its competitiveness and capable of preventing both cervical and anal cancer, is sold in over 130 countries.
Ahn-joon Choi, head of SK bioscience’s marketing division, said that “Through this agreement, we have secured a win-win strategy, a source of new growth in the vaccine market. In the future, we will expand the market and strengthen our position as a leading company by securing a comprehensive lineup of self-developed and jointly sold vaccines.”
SK chemicals’ subsidiary SK bioscience is strategically expanding as a global vaccine company, founded on its advanced vaccine R&D technologies and production capabilities.
Under the support of the Bill & Melinda Gates Foundation (BMGF), Phase 3 clinical trials for pediatric enteritis vaccines are being conducted with PATH (Program for Appropriate Technology in Health), a global health organization. With IVI (International Vaccine Research Institute), SK bioscience is preparing for product approval for a new typhoid vaccine. A next-generation pneumococcal vaccine in joint development with Sanofi Pasteur is currently undergoing Phase 2 clinical trials in the U.S.
In addition, SK bioscience’s self-developed vaccines for influenza and chickenpox obtained WHO PQ (Pre-qualification) certification in 2019 and are currently being exported worldwide. A technology export contract was also signed for the flu vaccine’s cell culture production technology to be applied to the “universal flu vaccine” developed by Sanofi Pasteur.
Recently, SK bioscience attracted attention after signing contract development and manufacturing agreements with domestic and foreign institutions such as the Ministry of Health and Welfare, the Ministry of Trade, Industry and Energy, BMGF, and CEPI (Coalition for Epidemic Preparedness Innovations) for the development of COVID-19 vaccines, while also signing agreements for the manufacturing of other COVID-19 vaccines developed by global companies.
SK bioscience plans to further strengthen its efforts to expand new businesses by △securing full-fledged overseas operations, △strengthening the R&D pipeline, and △expanding the business model.
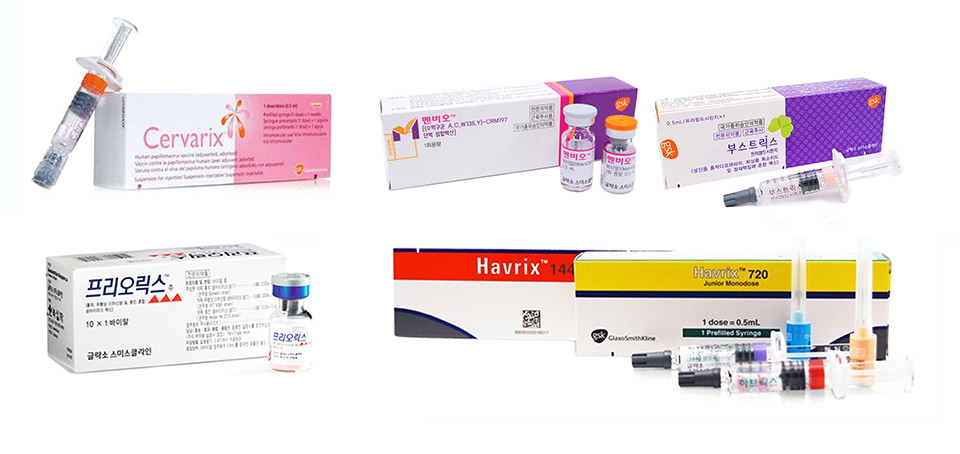
# Photo description: The five vaccines under the co-promotion contract by SK bioscience and GSK.